Sen Lab
- About
- Research
- People
- Publications
- Contact
Our lab focuses on research in the following areas:
To study epidermal homeostasis and tumor development, we established new methods to introduce specific combinations of genetic elements into 3-dimensionally intact human skin, containing human epidermal cells in the context of human dermal stroma and BM, regenerated on immune deficient mice. In this general approach, genetic elements are stably introduced via retroviruses to either knockdown or overexpress genes in primary human epidermal cells. Up to 10 alleles can be manipulated in this context. These cells are then combined with architecturally intact human dermis to regenerate human skin in the form of xenografts on immune deficient mice.
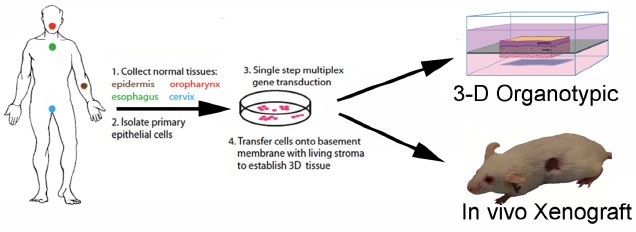
To model human epithelial tumorigenesis, inducible oncogenic RAS (generated by fusion with a mutant estrogen receptor ligand binding domain which can be activated by treatment with 4-hydroxytamoxifen) and Cdk4 (designed to mimic Rb pathway loss) is transduced into epidermal cells, which is then grafted onto immune deficient mice. Tumors that are formed using this system are clinically indistinguishable from spontaneous human squamous cell carcinomas. The inducible human cancer model permits study of temporally controlled tumor progression within architecturally intact human tissue. In this setting, the mechanistic basis for core cancer processes, such as the disruption of differentiation in the progression towards invasive neoplasia, can be dissected. Progression to invasion, the pathologic penetration of tissue boundaries by abnormal cells, is particularly amenable to study in new 3-dimensionally intact human skin models because of the presence of key basement membrane stromal elements and organized tissue architecture.
Summary
The key advantages of MFG:
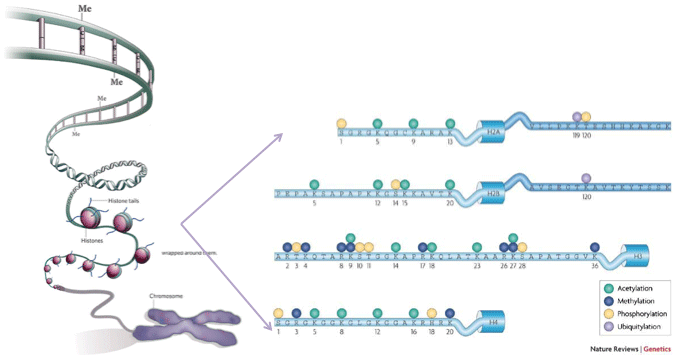
We are interested in determining the epigenetic factors that regulate epidermal homeostasis and progression to neoplasia. The term epigenetics refers to the transmission of heritable information from parental to daughter cells distinct from the primary DNA sequence.
The two major epigenetic mechanisms involve the covalent modification of either DNA itself or histones that wrap around the DNA.